- Joined
- Dec 3, 2009
- Messages
- 7,138
- Reaction score
- 12,355
- Age
- 59
Offline
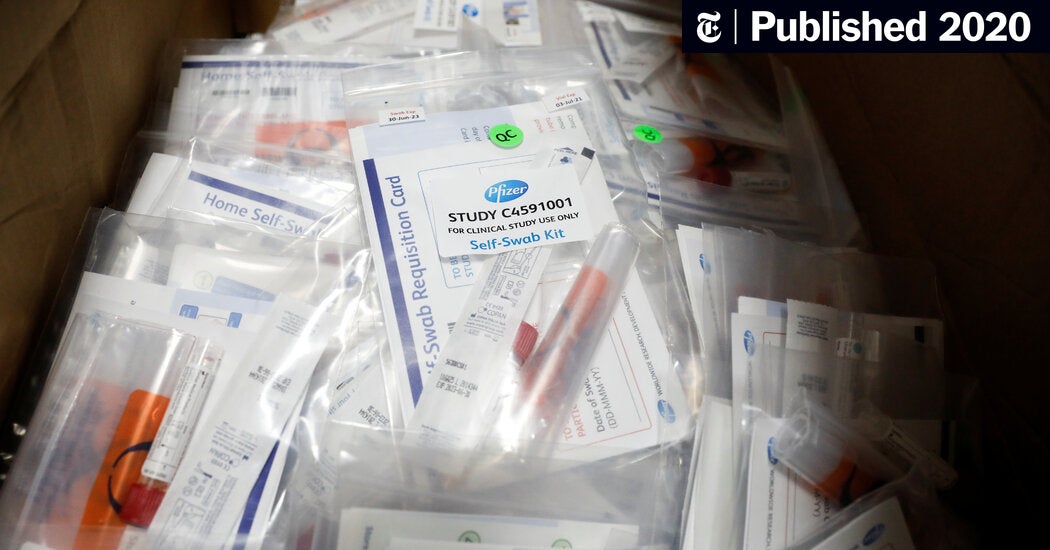
Looks like third week of November now for Pfizer. They may have efficacy data by the end of October, but need more time to gather enough safety and manufacturing data to apply for approval. FDA came out with new guidelines very recently. That probably had an impact. So long as it works and is safe, need some vaccines out there soon. The spike in cases in the USA and Europe is real and steep.
Pfizer Says It Won’t Seek Vaccine Authorization Before Mid-November
"Friday’s announcement represents a shift in tone for the company and its leader, who has repeatedly emphasized the month of October in interviews and public appearances.
In a statement posted to the company website, the chief executive, Dr. Albert Bourla, said that although Pfizer could have preliminary numbers by the end of October about whether the vaccine works, it would still need to collect safety and manufacturing data that will stretch the timeline to at least the third week of November."
 www.nytimes.com
www.nytimes.com
Pfizer Says It Won’t Seek Vaccine Authorization Before Mid-November
"Friday’s announcement represents a shift in tone for the company and its leader, who has repeatedly emphasized the month of October in interviews and public appearances.
In a statement posted to the company website, the chief executive, Dr. Albert Bourla, said that although Pfizer could have preliminary numbers by the end of October about whether the vaccine works, it would still need to collect safety and manufacturing data that will stretch the timeline to at least the third week of November."

Pfizer Says It Won’t Seek Vaccine Authorization Before Mid-November (Published 2020)
Friday’s announcement represents a shift in tone for the company and its leader, who has repeatedly emphasized the month of October in interviews and public appearances.




